How it works.
Your typical glowstick consists of a plastic tube filled with hydrogen peroxide, or H2O2. There is a small glass tube inside that is filled with phenyl oxalate esters, or bis(2,4,6-trichlorophenyl)oxalate (TCPO). When you bend the glow stick, the little tube breaks and the chemicals mingle. The electrons in the dye begin to get excited and move up. The result is a glow.
Reaction type
Glow sticks involve what is referred to as a "redox", aka reduction/oxidization reaction. The mechanism for the reaction is what we refer to as chemiluminescence, or a way of producing light chemically.Welsh, Emma. “What Is Chemiluminescence?” Science in School, EIROforum, 16 Dec. 2022, www.scienceinschool.org/article/2011/chemiluminescence/.
For more information, check out the following:
http://www.compoundchem.com/2014/10/14/glowsticks/
For more information, check out the following:
http://www.compoundchem.com/2014/10/14/glowsticks/
Word equation
Hydrogen peroxide + TCPO [also known as bis(2,4,6-trichlorophenyl)oxalate] + dye ------>>>> trichlorophenol + carbon dioxide + excited dye.
Chemical Equation
H2O2(aq) + C14H10O4(aq) + dye -> C6H2Cl3OH + 2CO2 + excited dye
Assume bis(2,4,6-trichlorophenyl)oxalate as the solvent, and select a dye from below.
for example, H2O2(aq) + C14H10O4(aq) + (dye from below) = C6H2Cl3OH + 2CO2 + x
solve for x.
Assume bis(2,4,6-trichlorophenyl)oxalate as the solvent, and select a dye from below.
for example, H2O2(aq) + C14H10O4(aq) + (dye from below) = C6H2Cl3OH + 2CO2 + x
solve for x.
Explanation of “dye”
Edit, 3/21/23:
On the definition of “dye”
Multiple different kinds of fluorescent dye with different chemical equations are used for glowsticks.
The type of dye ultimately depends on what colour you want to use. “Dye” in this chemical equation can be substituted for the chemical formula of whatever dye you are using. Substitute a color from the below chart to calculate the equation. Pick your favourite color.
For example: if you want 9,10-diphenylanthracene: C26H18
H2O2(aq) + C14H10O4(aq) + C26H18 -> C6H2Cl3OH + 2CO2 + excited dye
An article at thoughtCo suggests the following types (Helmenstine, Anne Marie, Ph.D. "How Glow Stick Colors Work." ThoughtCo, Feb. 16, 2021, thoughtco.com/how-glow-stick-colors-work-4064535.)
On the definition of “dye”
Multiple different kinds of fluorescent dye with different chemical equations are used for glowsticks.
The type of dye ultimately depends on what colour you want to use. “Dye” in this chemical equation can be substituted for the chemical formula of whatever dye you are using. Substitute a color from the below chart to calculate the equation. Pick your favourite color.
For example: if you want 9,10-diphenylanthracene: C26H18
H2O2(aq) + C14H10O4(aq) + C26H18 -> C6H2Cl3OH + 2CO2 + excited dye
An article at thoughtCo suggests the following types (Helmenstine, Anne Marie, Ph.D. "How Glow Stick Colors Work." ThoughtCo, Feb. 16, 2021, thoughtco.com/how-glow-stick-colors-work-4064535.)
- Blue: 9,10-diphenylanthracene (Carbon26Hydrogen18)
- Blue-Green: 2-chloro-9,10-diphenylanthracene (Carbon30Hydrogen18))
- Green: 2-Chloro-9,10-bis(phenylethynyl)anthracene (Carbon30Hydrogen17Chlorine)
- Yellow-Green: 1-Chloro-9,10-bis(phenylethynyl)anthracene (
- Yellow: 1-chloro-9,10-bis(phenylethynyl)anthracene
- Yellow: 1,8-dichloro-9,10-bis(phenylethynyl)anthracene
- Orange-Yellow: Rubrene (C42H28)
- Orange: 5,12-bis(phenylethynyl)-naphthacene (?) or Rhodamine 6G (C28H31N2O3Cl)
- Red: 2,4-di-tert-butylphenyl 1,4,5,8-tetracarboxynaphthalene diamide (?) or Rhodamine B (C28H31ClN2O3)
- Infrared: 16,17-dihexyloxyviolanthrone, 16,17-butyloxyviolanthrone, 1-N,N-dibutylaminoanthracene, or 6-methylacridinium iodide
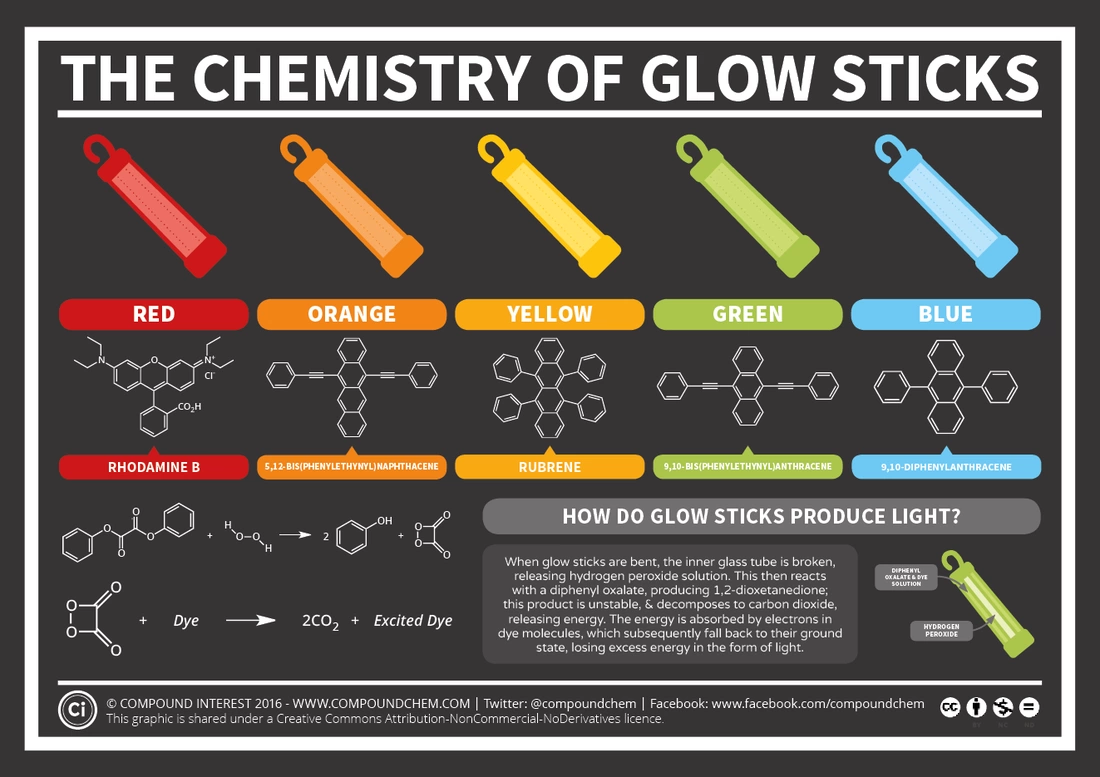
Here is a chart from the website CompoundChem, on their page The Chemistry of Glow Sticks. The chart is under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
(http://www.compoundchem.com/2014/10/14/glowsticks/)
(http://www.compoundchem.com/2014/10/14/glowsticks/)